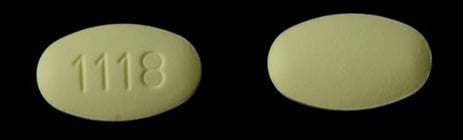
Blood-pressure drugs containing Losartan recalled due to potential carcinogen contamination - National | Globalnews.ca

Teva Pharmaceuticals USA, Inc. Issues Voluntary Nationwide Recall of Losartan Potassium 25 mg and 100 mg Tablets USP, Sold Exclusively to Golden State Medical Supply | FDA